Conductivity Testers
GAO’s water conductivity testers are devices that are used to measure the electrical conductivity of water. Conductivity is a measure of how well a solution can conduct electricity, and in water, this property is determined by the concentration of dissolved ions, such as salts and minerals. The meter may be a simple handheld device or a more advanced instrument with features such as automatic temperature compensation, data logging, and multiple ranges of measurement. High levels of conductivity in water can indicate the presence of dissolved salts and minerals which may be harmful to aquatic life or may affect the taste and quality of drinking water.
GAO’s water conductivity testers are composed of electrodes, circuitry, display, power source, calibration solutions, meter, and carrying case.
The other common names of conductivity testers are conductivity meters, electrical conductivity analyzers, conductance meters, salinity testers, total dissolved solids meters, electrolyte conductivity testers, ion concentration analyzers, cell constant meters, dissolved ion concentration meters, electrical conductance probes, solution conductivity sensors, ionic strength meters, electrolyte concentration analyzers, conductivity measurement instruments and specific conductance meters.
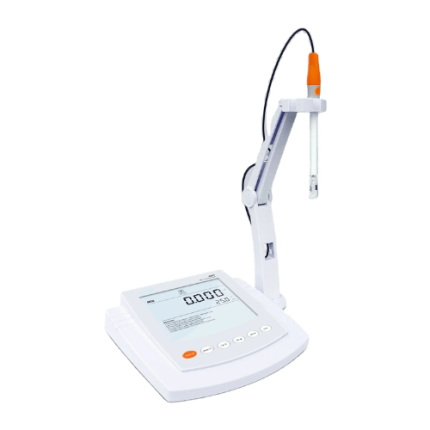
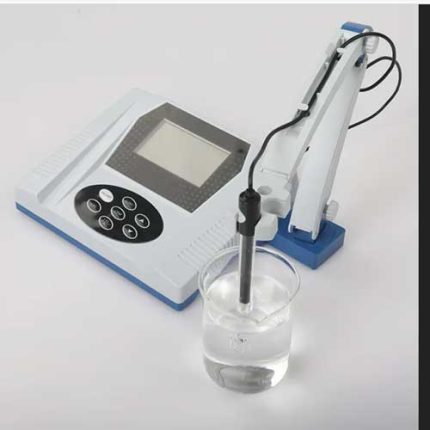
Benchtop pH/Conductivity Meter
GAOTek Accurate Digital Conductivity
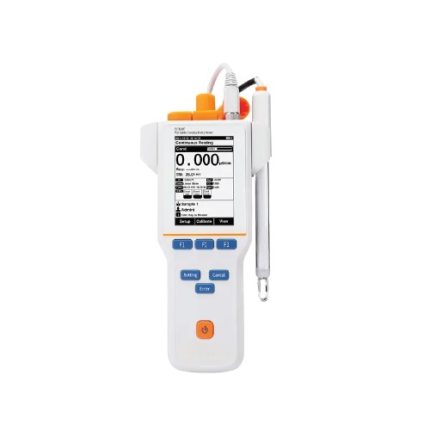
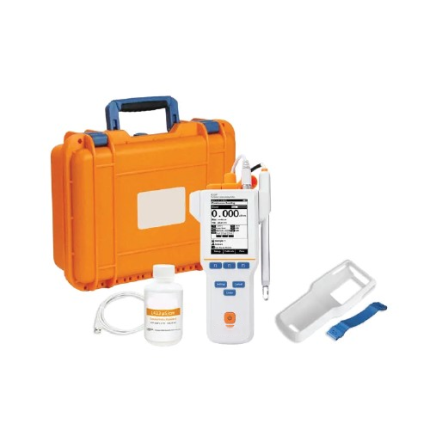
GAOTek Advanced Portable Lab Conductivity Meter
GAOTek Affordable Fuel Oil Conductivity Tester
GAOTek Automatic Conductivity Meter
GAOTek Automatic Conductivity Tester
GAOTek Automatic Conductivity Tester
GAOTek Automatic Portable Water Conductivity Meter
GAOTek Automatic Temperature Compensation
GAOTek Automatic Temperature Compensation Conductivity
GAOTek Automatic Temperature Water Conductivity Meter
GAOTek Battery Cell Conductance Tester
GAOTek Battery Conductance Tester
GAOTek Battery Conductivity Tester
GAOTek Bench Top Conductivity Tester
GAOTek Benchtop Conductivity Ash Meter
GAOTek Benchtop Conductivity Meter
GAOTek Benchtop Conductivity Meter
GAOTek Benchtop Conductivity Meter
GAOTek Benchtop Conductivity Meter
GAOTek Benchtop Conductivity Meter For Water Quality Analysis
GAOTek Benchtop Conductivity Meter With Temperature Compensation
GAOTek Benchtop Conductivity Salinity Temperature Meter
GAO Tek’s water conductivity testers have the following functions:
- Determining water purity- GAO water conductivity testers can be used to measure the purity of water by detecting the presence of dissolved salts and minerals that can affect the taste and quality of the water.
- Monitoring water quality- In environmental monitoring applications, they are used to monitor water quality and detect any changes that may indicate pollution or other environmental factors.
- Controlling water treatment processes- Our water conductivity testers are used in water treatment plants to control the concentration of dissolved salts and minerals in the water, which can affect the efficiency of treatment processes.
- Ensuring the safety of aquatic life- High levels of conductivity in water can be harmful to aquatic life, and our water conductivity testers are used to monitor water quality in aquariums and other aquatic environments to ensure the safety of the animals.
- Conducting research- GAO’s water conductivity testers are also used in research applications to study the properties of water and its interaction with other substances.
- Detecting contaminants- GAO water conductivity testers can also be used to detect the presence of contaminants in water. Some contaminants, such as heavy metals and organic compounds, can affect the electrical conductivity of water.
GAO’s water conductivity testers are rugged, versatile, and precision measurement instruments. They comply with applicable industry standards such as ASTM D1125, ISO 7888, EPA Method 120.1, and EN 27888.
Our water conductivity testers help our customers comply with U.S. government regulations such as:
- Safe Drinking Water Act (SDWA)
- Clean Water Act (CWA)
- Environmental Protection Agency
Our water conductivity testers help our clients in complying with relevant Canadian government regulations such as:
- Canadian Environmental Protection Act (CEPA)
- Fisheries Act
- Provincial and territorial regulations
GAO’s water conductivity testers have the following applications:
- Water treatment-GAO water conductivity testers are used in water treatment plants to monitor the quality of water and ensure that it meets regulatory standards. This includes testing the conductivity of raw water, as well as monitoring the effectiveness of water treatment processes such as reverse osmosis (RO) and deionization.
- Aquaculture- In aquaculture operations, water conductivity testers are used to monitor the quality of water in fish tanks, ponds, and other aquatic environments. This includes testing the water’s conductivity, as well as monitoring other water quality parameters such as pH, dissolved oxygen, and temperature.
- Swimming pools and spas- Our water conductivity testers are used to monitor the quality of water in swimming pools and spas. This includes testing the conductivity of the water to ensure that it is safe for swimmers and monitoring other water quality parameters such as pH and chlorine levels.
- Environmental monitoring- Our water conductivity testers are used in environmental monitoring applications to test the quality of water in rivers, lakes, and other bodies of water.
- Laboratory research- GAO water conductivity testers can be used in laboratory research to measure the conductivity of water and other aqueous solutions.
Our water conductivity testers can be shipped overnight within the continental U.S. and Canada, and can be shipped globally.